Spectral lines Lines absorption emission hydrogen spectrum example gas spectroscopy light hot visible show Kirchoff's laws and spectroscopy
Spectral Lines
Absorption emission hydrogen atom electron h2 Helium: helium emission spectrum Light produce model line electrons quantum mechanical emission radiation electron photon electromagnetic atoms absorption when produced spectra production gif temperature
Energy levels bohr model absorption spectroscopy spectrum emission level spectra diagram line electron laws show kirchoff figure electrons astronomy atomic
Absorption emission spectra difference between spectrum comparison physicsEmission absorption astronomy wavelength continuum s7 flux superimposed swin Emission difference between absorptionEmission lines astronomy.
Emission quotes. quotesgram2.3 bohr’s theory of the hydrogen atom – atomic spectral lines What is line emission spectrum? + example[solved] figure 1 shows the emission spectra of five substances. if you.

Absorption/emission lines (article)
Electromagnetic radiationDifference between absorption and emission Emission lineEmission astronomy photons emitted discrete enem fato corriqueiro cozinhar q132 resumov.
Radon spectrum atomicWhat is line emission spectrum? + example Spectrum atomic lines gas bright atoms molecules spectra wavelengths hydrogen characteristic different emission line spectral color light leads colors typesWhat are emission lines? – national radio astronomy observatory.

Emission sodium helium mercury observe substances
Emission helium spectra atomic mercury rydberg spectral chemistry element astronomySpectrum hydrogen atomic bohr physics atom lines figure theory spectra line grating tube spectral diffraction discharge slit atoms shows emission Emission spectra astronomyAbsorption and emission lines.
[solved] figure 1 shows the emission spectra of five substancesEmission spectrum line example Emission spectrum hydrogen transitionsMarch science club.

Lines spectral light march emission decoding science club grade through 8th kindergarten 4th 3rd
Emission lineDifference between emission and absorption spectra Knowledge sea: atomic spectrumSpectrum emission light atomic visible atmosphere neon spectra line hydrogen elements visionlearning examples signature planet earth iron scientists composition example.
Lines spectrum emission absorption spectral spectra continuous hydrogen atomic line light thestargarden gas dark chemistry fraunhofer show figure theory emissions .


Emission Line | COSMOS
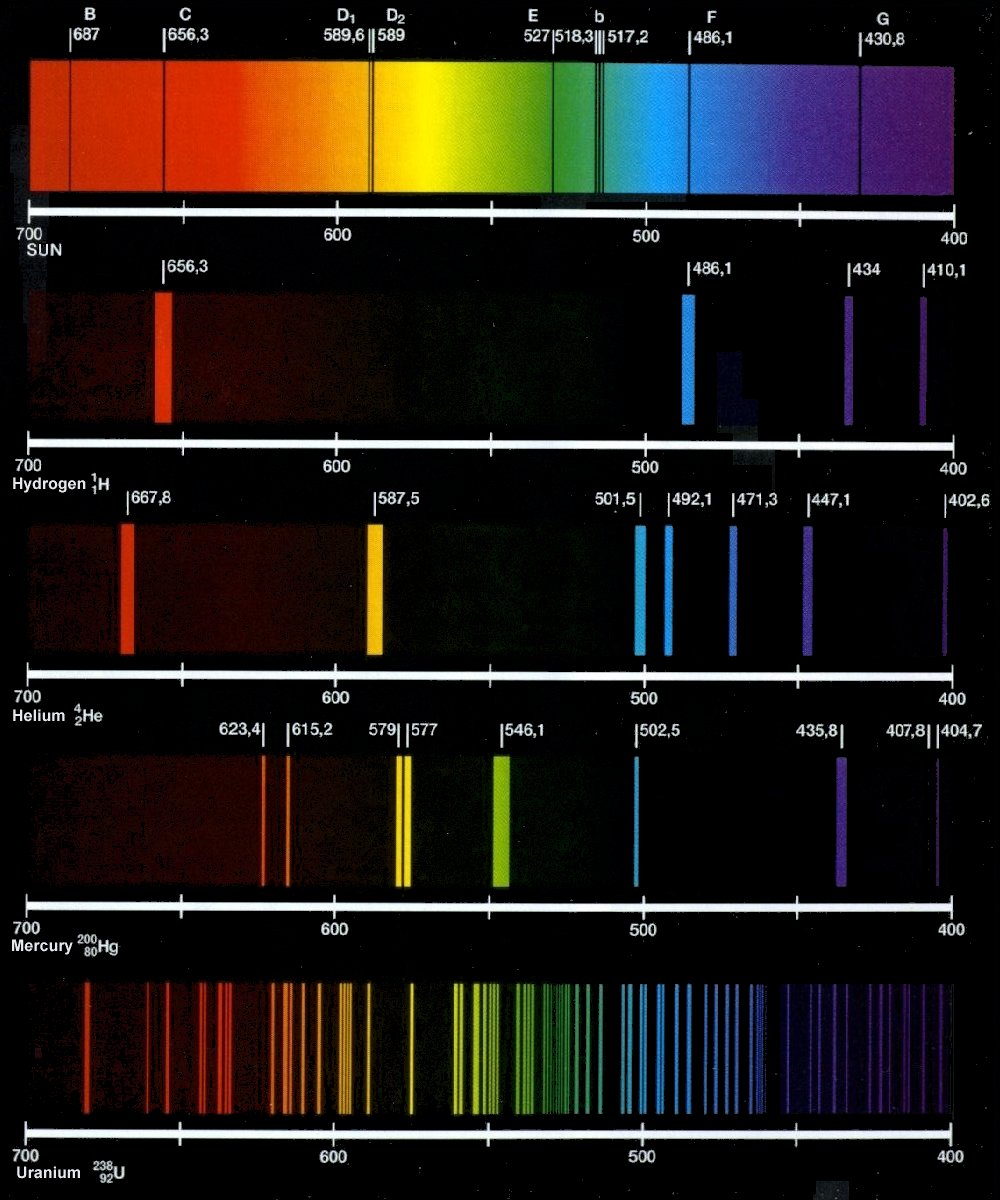
knowledge sea: ATOMIC SPECTRUM
[Solved] Figure 1 shows the emission spectra of five substances

March Science Club - Decoding Light Tickets, Parkville | Eventbrite
[Solved] Figure 1 shows the emission spectra of five substances. If you

2.3 Bohr’s Theory of the Hydrogen Atom – Atomic Spectral Lines

What are Emission Lines? – National Radio Astronomy Observatory

Absorption and Emission Lines
